The development of a wearable neuromorphic electronic device platform in the form of an electronic fiber capable of biosignal recognition and learning
Results achieved by the team led by Professor Wang, Gun-uk are published in the world-renowned academic journal, Science Advances.

▲ From the left, Professor Wang, Gun-uk (corresponding author),
Ham, Seong-gil, and Jang, Seong-hoon (co-first authors)
A neuromorphic chip is a next-generation computing device technology for processing unstructured large-capacity data at high speed and using low power and is achieved by mimicking the way synapses transmit information between neurons to simultaneously perform both memory operations and computation. The incorporation of AI hardware technology for biosignal diagnostics into healthcare systems is an innovative technology that is a key driver of the fourth industrial revolution, and demand for it continues to increase.
The teams led by Professor Wang, Gun-uk of KU-KIST Graduate School of Converging Science and Technology and Professor Kim, Tae-wook of Jeonbuk National University jointly developed an AI hardware basic technology that can be used in E-textile next-generation healthcare systems. A highly stable one-dimensional artificial synaptic device that can be woven like fabric was developed and integrated to successfully determine and diagnose a representative biosignal, an electrocardiogram (ECG) signal.
The platform developed by the research team is a method for manufacturing multi-synapse devices on a thin Ag (silver) wire (diameter: 100 μm) as thin as a hair. It is composed of a ferroelectric-based three-terminal structure that can store electrical information in several stages by stacking an organic ferroelectric (PVDF-TrFE) and the organic semiconductor pentacene, which is used as a channel. The ability to change dielectric polarization according to the transmitted electrical signal is a key characteristic of the ferroelectric and plays a role in changing the conductivity of the artificial synaptic device according to electrical stimulation. The technology mimics neurotransmitters responding to inter-neuron stimulation in the brain and the memory-related functions thereby performed by synapses.
The artificial synapse developed by the research team has stably implemented synaptic plasticity, which is a key characteristic of aspects of memory functioning, such as the long-term strengthening of memory storage and the long-term suppression of memory, and achieved through regulation of the strength of the synaptic connections between neurons. It was confirmed that these properties were maintained even under very harsh mechanical deformation. In addition, structurally, it is more advantageous to apply neuromorphic technology than other previously reported textile-type devices; an artificial synaptic device that is multiply mounted on a silver wire serving as a gate in a 3-terminal structure can be woven in an e-textile form like a fabric, with separate silver wires acting as source and drain. It can easily be configured as a neuromorphic computational circuit, a NOR-type artificial synaptic array. This method can solve the problem of interference between adjacent cells due to the current leakage associated with existing fabric type structures and the additional circuit configuration requirements of artificial neural network circuit construction. It is expected to be an advantageous platform technology when applied to healthcare devices where wearability in a fabric-type form is important.
The research team conducted learning on five types of electrocardiograms (ECGs), one normal-state heartbeat type and four arrhythmia types, based on the electrical characteristics of the artificial synaptic devices for biosignal learning and cognitive testing. The artificial brain neural network model composed of artificial synaptic devices demonstrated 70% accuracy in this arrhythmia diagnosis simulation. Based on these results, Professor Wang said of the device, “It is a core technology for the next-generation healthcare field using neuromorphic technology, and shows potential for the development of applications that can analyze biological signals from the body in real time and inform of signs of abnormality.”
The result of this study is expected to be the cornerstone of the development of wearable-based neuromorphic hardware technology and the development of next-generation intelligent healthcare systems. In recognition of its significance, the research was published in the July 11 edition of Science Advances (impact factor: 13.116), the sister publication of the international journal Science.
This work was supported by the National Research Foundation of Korea (NRF-2019R1A2C2003704, 2019R1A6A3A01095700, and 2020R1A2C2010163), the KU-KIST Research Fund, a Korea University Grant, and Samsung Electronics Strategic Industrial-Academic Project .
* Paper title : One-dimensional organic artificial multi-synapses enabling electronic-textile neural network for wearable neuromorphic applications
* Author information : Ham, Seong-gil (Korea University, first author), Kang, Min-ji (Jeonbuk National University, co-first author), Jang, Seong-hoon (Korea University, co-first author), Jang, Jin-gon (Korea University, co-author), Choi, Sang-hyeon (Korea University, co-author), Kim, Tae-wook (Jeonbuk National University, co-corresponding author), Wang, Gun-uk (Korea University, Corresponding author)

▲ Figure description: (A) Schematic diagram of one-dimensional artificial synapse, (B) Artificial synaptic device image (top) and photo (bottom) enlarged using a scanning electron microscope (SEM), (C) Electrical characteristic that mimics synapses for which the conductivity of the device changes according to the electrical pulse signal, (D) The driving principle of synaptic function simulation. The conductivity of the device is changed by inducing polarization of the ferroelectric according to the voltage applied to the device.
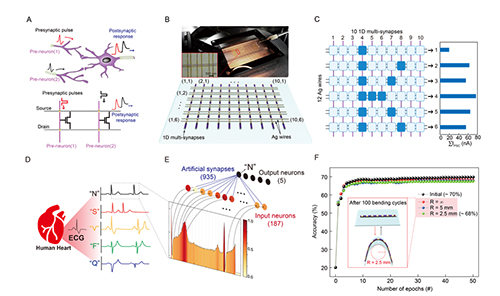
▲ Figure description: (A) Schematic diagram of signal transmission through parallel connection of neurons and synapses between neurons (top) and artificial synaptic array structure (NOR-type), which is a neuromorphic computing circuit (bottom), (B) Structure and schematic diagram of parallel type artificial synapse array connected with the produced artificial synapse, (C) The ‘k“ pattern encoded by learning in an artificial synapse array, (D) Normal heartbeat ECG and 4 arrhythmia ECGs used in the simulation, (E) Artificial neural network model for learning ECG type, (F) Results of the ECG pattern learning test showing a recognition rate of 70%, with no significant decrease in recognition rate apparent even after 100 cycles on the bend tester.



